Cosmic Kidney Disease
an integrated pan-omic, physiological and morphological study into spaceflight-induced renal dysfunction
2/6/20254 min read


Missions into Deep Space are planned this decade. Yet the health consequences of exposure to microgravity and galactic cosmic radiation (GCR) over years-long missions on indispensable visceral organs such as the kidney are largely unexplored.
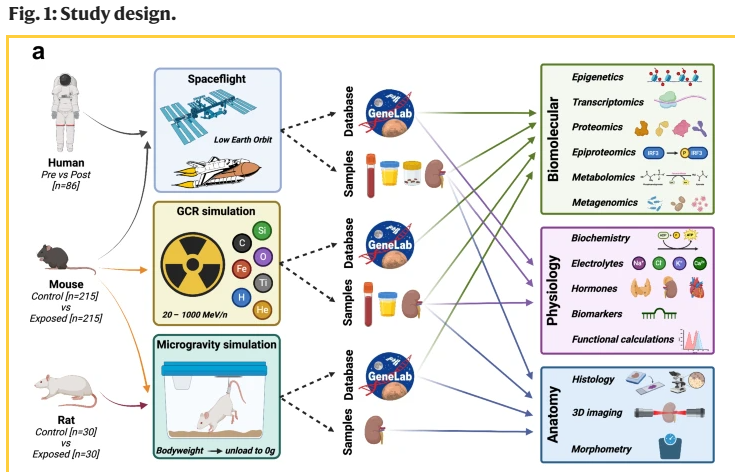
We performed biomolecular (epigenomic, transcriptomic, proteomic, epiproteomic, metabolomic, metagenomic), clinical chemistry (electrolytes, endocrinology, biochemistry) and morphometry (histology, 3D imaging, miRNA-ISH, tissue weights) analyses using samples and datasets available from 11 spaceflight-exposed mouse and 5 human, 1 simulated microgravity rat and 4 simulated GCR-exposed mouse missions.
We found that spaceflight induces:
1) renal transporter dephosphorylation which may indicate astronauts’ increased risk of nephrolithiasis is in part a primary renal phenomenon rather than solely a secondary consequence of bone loss;
2) remodeling of the nephron that results in expansion of distal convoluted tubule size but loss of overall tubule density;
3) renal damage and dysfunction when exposed to a Mars roundtrip dose-equivalent of simulated GCR.
The renewed zeitgeist for space travel, ushered in by the advent of commercial spaceflight and tourism, has spurred on state-funded agencies to embark on even more ambitious exploratory ‘Deep Space’ missions.
The first of these, namely the Artemis Program, Lunar Gateway space station and later the Deep Space Transport/Mars Missions, will for the first-time place humans outside of Earth’s protective magnetic field exposed to significant quantities of unmitigated space radiation and weightlessness for many months to years at a time.
The health effects of low Earth orbit (LEO) spaceflight (e.g. the International Space Station) are multiple, with much of the research communities’ intense focus centered on the musculoskeletal, neurological, ocular and cardiovascular degeneration that can manifest as early as a few weeks into a mission.
Notably, the effects of LEO spaceflight on many other organ systems are less clear, with indispensable organs such as the kidneys receiving relatively little attention in the absence of overt symptoms.
However, in the face of extended periods of deep space travel health issues may only present with late onset due to cloaked subclinical pathophysiology and chronic damage eating into the extensive functional reserves of such organs.
The kidneys are critical in regulating blood pressure, by controlling both the renin/angiotensin/aldosterone system (RAAS) and the reabsorption of sodium chloride and water from the urine.
In microgravity, the cephalad redistribution of blood volume with the concomitant decrease in the pressure gradient should reduce kidney perfusion. In turn, a lower perfusion pressure within the renal artery should lead to a greater release of renin and activation of the RAAS.
However, studies show a marked reduction of diuresis and natriuresis in space with the RAAS, and antidiuretic hormone (ADH) increasing through unknown mechanisms1,2,3.
These unexplained effects, be they mediated through known homeostatic mechanisms, or more theoretical ones, such as tensegrity4, are germane to kidney stone formation (i.e. nephrolithiasis).
Astronauts have an unusually high rate of kidney stone formation, with 1-year post-flight astronauts experiencing incidence rates of 2–7 times that of pre-flight estimates, and in-flight risk estimated to be double that again5.
This is of mission critical significance, one Soviet in-flight renal stone episode nearly caused a mission termination due to the severe symptoms, but was relieved by spontaneous stone passage by the cosmonaut just before an urgent deorbit was initiated6.
It has been demonstrated that spaceflight associated changes in urinary biochemistry favor kidney stone formation7,8,9.
Microgravity may have a direct effect on the crystallization and nucleation of nascent kidney stones10, but this is dwarfed by the net biochemical urinary changes historically attributed to the microgravity-induced bone demineralization observed in spaceflight.
A school of thought that has led to a paucity of research into other pathophysiological phenomena that may be contributing to the unusually high incidence of nephrolithiasis.
The kidney is a plastic organ and is capable of dynamically remodeling the architecture of the nephron in response to changes in blood pressure or dietary potassium.
An 18 day potassium-free diet resulted in a 31% increase in rat kidney weight, which was mainly seen in the medullary collecting duct11.
Changes in the size of nephron segments in wild type mice have been observed using advanced imaging techniques in a period as short as three days12.
So, it is plausible that spaceflight-induced shifts in fluid and electrolyte handling could precipitate maladaptive remodeling events that contribute to the underlying pathophysiology.
In addition to the well-studied environmental stressor of microgravity, deep space missions beyond the protection of the Geomagnetosphere (the Van Allen belts) are exposed to space radiation.
Space radiation comprises both intermittent events (e.g. Solar Particle Events or Solar Proton Events) with the most energetic being a Coronal Mass Ejection and Solar Wind and Galactic Cosmic Radiation (GCR), with GCR having the most significant health impacts.
There is concern regarding the carcinogenic effect of GCR exposure in the planned Mars Missions.
However, the kidney is an exquisitely radiation sensitive organ; it is the dose limiting organ in abdominal radiotherapy13.
Chronic kidney dysfunction can occur with acute low linear energy transfer (LET) γ-radiation doses as low as <0.5 Gy14, the LET dose expected on a Mars Mission.
However, GCR comprises low-LET x- and γ-rays with protons and high energy ‘HZE’ ions of heavier elements (e.g. silicon, titanium, iron). It has been established that exposure to HZE ions results in mitochondrial damage15,16.
The renal proximal tubule is heavily dependent on mitochondrial respiration17 and is one of the main tubular segments in which damage is associated with kidney failure18.
Indeed, proximal tubular injury is a prominent histological feature in medical (low-LET) radiation nephropathy19.
Due to their dependence on mitochondrial aerobic respiration, proximal tubular cells are extremely vulnerable to mitochondrial damage that we would expect from HZE exposure in addition to low-LET radiation damage.
We hypothesized that the biochemical urinary changes seen in spaceflight might reflect functional and morphological changes detectable in the kidney secondary to microgravity, possibly due to abnormal renal perfusion.
Further, we hypothesized that GCR exposure will cause mitochondrial and therefore proximal tubular dysfunction, leading to tissue damage and potentially irreversible loss of renal function, in addition to the known micro and macrovascular damage caused by GCR.
In order to explore these questions, we took a pan-omics approach incorporating anatomical and physiological parameters from 20 independent human and rodent spaceflight missions/simulations that had extant data and/or samples from which we could generate novel data (Fig. 1).
Exposing human colonization of the Moon and Mars as Space Fraud, Waste and Abuse
© 2025. All rights reserved. SpaceFraud.Tech
